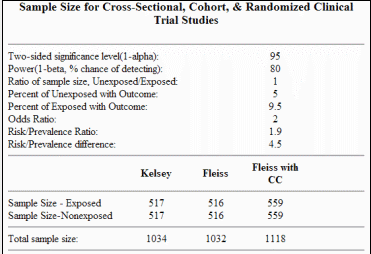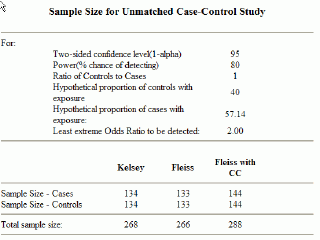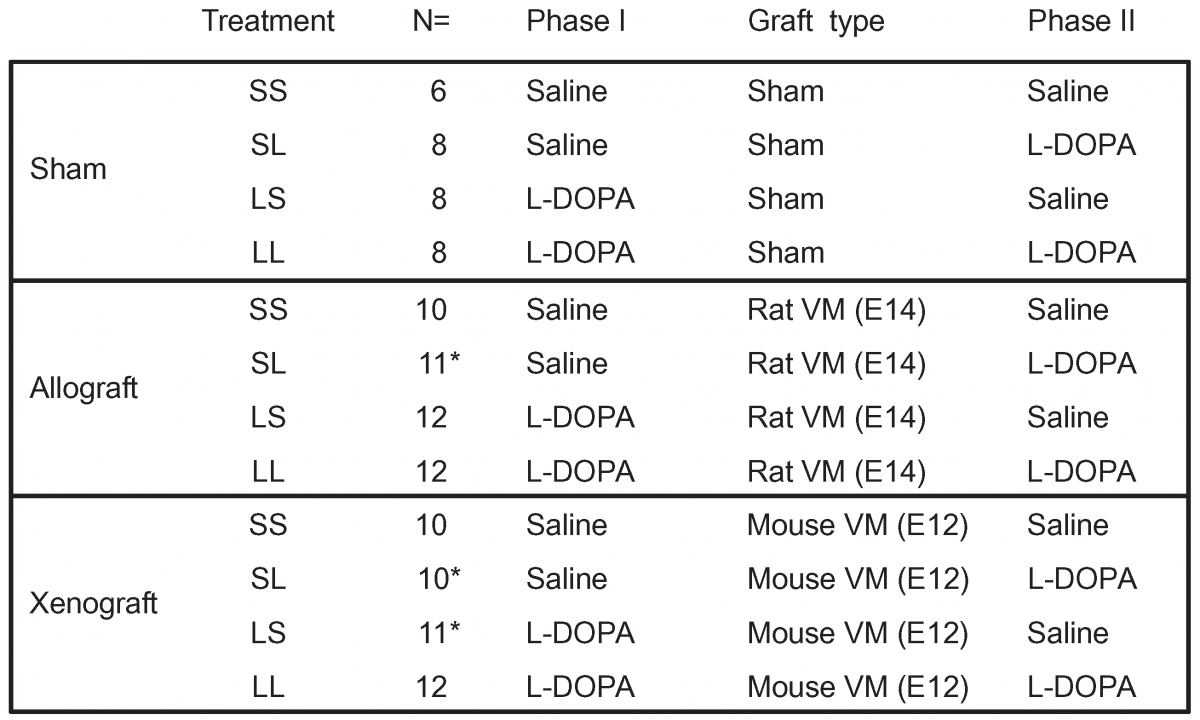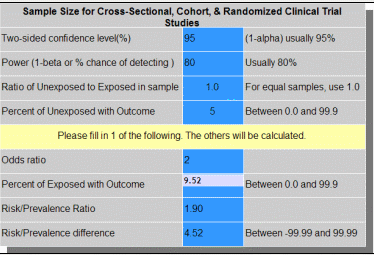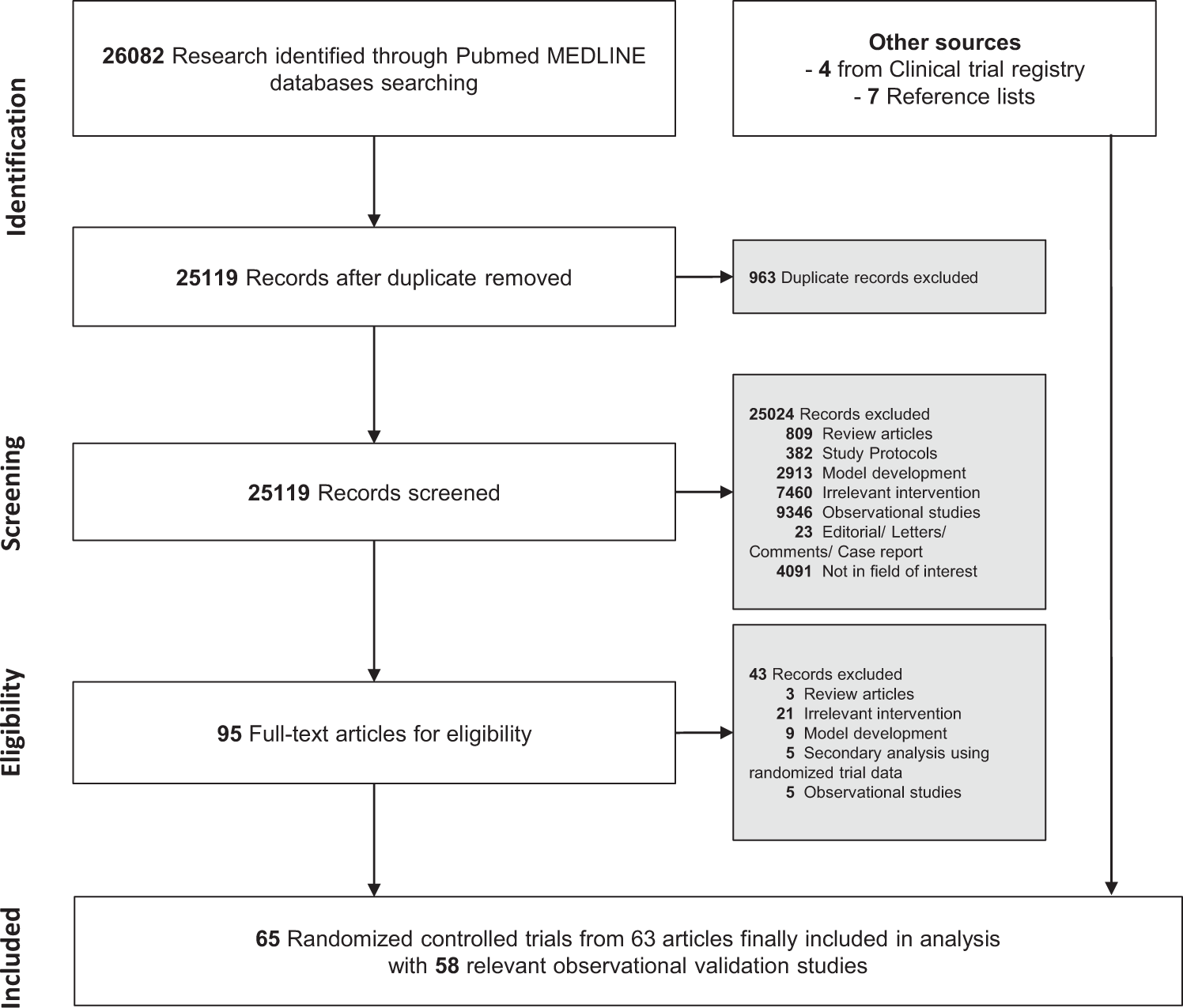
Clinical impact and quality of randomized controlled trials involving interventions evaluating artificial intelligence prediction tools: a systematic review | npj Digital Medicine

Sample size re-estimation in a superiority clinical trial using a hybrid classical and Bayesian procedure | Semantic Scholar
Resource use, costs, and approval times for planning and preparing a randomized clinical trial before and after the implementation of the new Swiss human research legislation | PLOS ONE
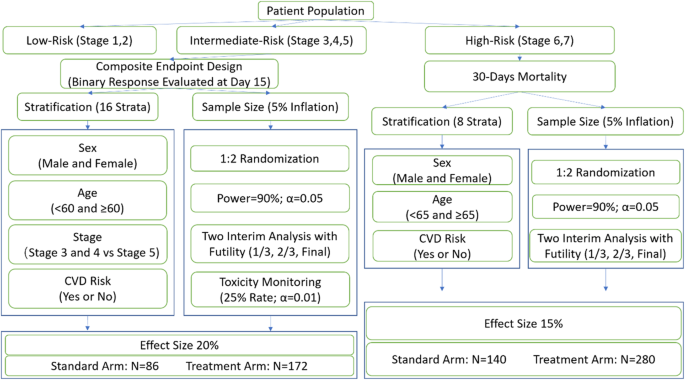
Statistical design of Phase II/III clinical trials for testing therapeutic interventions in COVID-19 patients | BMC Medical Research Methodology | Full Text

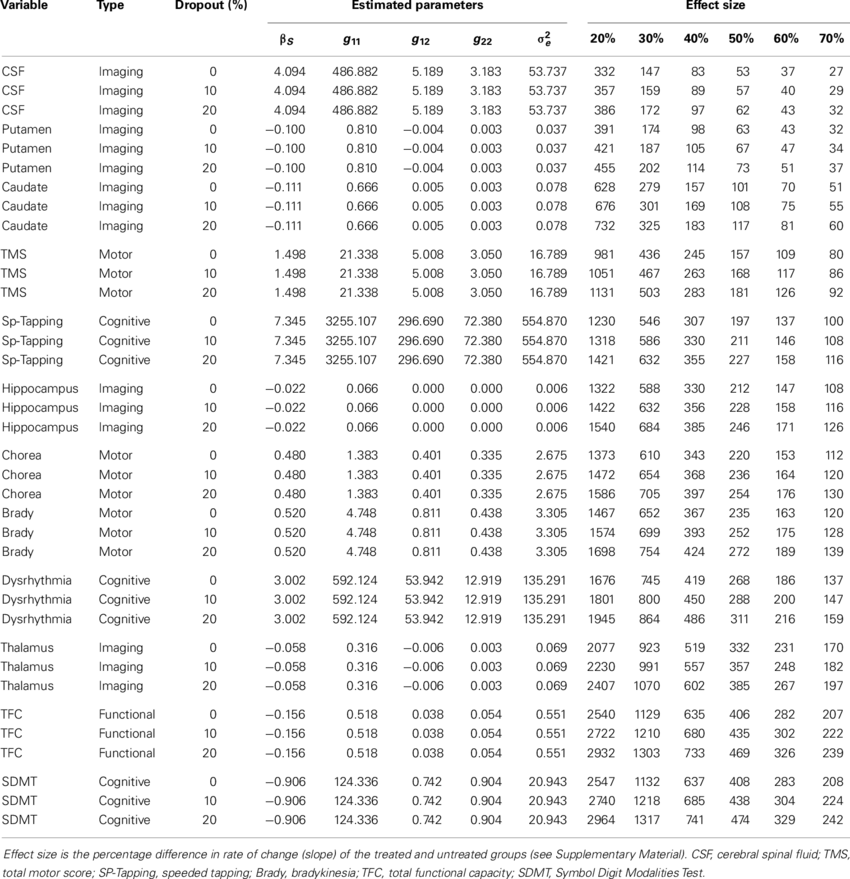
Table 2 from Power and Sample Size Calculations in Clinical Trials with Patient-Reported Outcomes under Equal and Unequal Group Sizes Based on Graded Response Model: A Simulation Study. | Semantic Scholar

Table 4 from Power and Sample Size Calculations in Clinical Trials with Patient-Reported Outcomes under Equal and Unequal Group Sizes Based on Graded Response Model: A Simulation Study. | Semantic Scholar
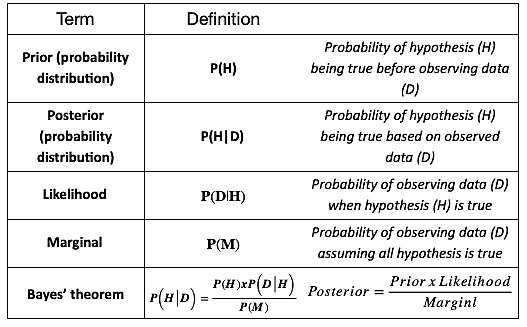
Bayesian approach for sample size estimation and re-adjustment in clinical trials – Biostatistics & Bioinformatics Services

Sample Size Tables for Clinical Studies: 9781405146500: Medicine & Health Science Books @ Amazon.com
Use of High-Frequency In-Home Monitoring Data May Reduce Sample Sizes Needed in Clinical Trials | PLOS ONE

Generalized Competing Event Models Can Reduce Cost and Duration of Cancer Clinical Trials | JCO Clinical Cancer Informatics
Sample Size in Statistics (How to Find it): Excel, Cochran's Formula, General Tips - Statistics How To

Sample Sizes For Clinical, Laboratory And Epidemiology Studies - 4th Edition By David Machin & Michael J Campbell & Say Beng Tan & Sze Huey Tan : Target

Table 3 from Power and Sample Size Calculations in Clinical Trials with Patient-Reported Outcomes under Equal and Unequal Group Sizes Based on Graded Response Model: A Simulation Study. | Semantic Scholar

Sample Size Tables for Clinical Studies: 9781405146500: Medicine & Health Science Books @ Amazon.com

![PDF] Sample size estimation in diagnostic test studies of biomedical informatics | Semantic Scholar PDF] Sample size estimation in diagnostic test studies of biomedical informatics | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/596c4b9e0b7817120acb917cff4728fab9f95ca8/4-Table2-1.png)

.jpg)