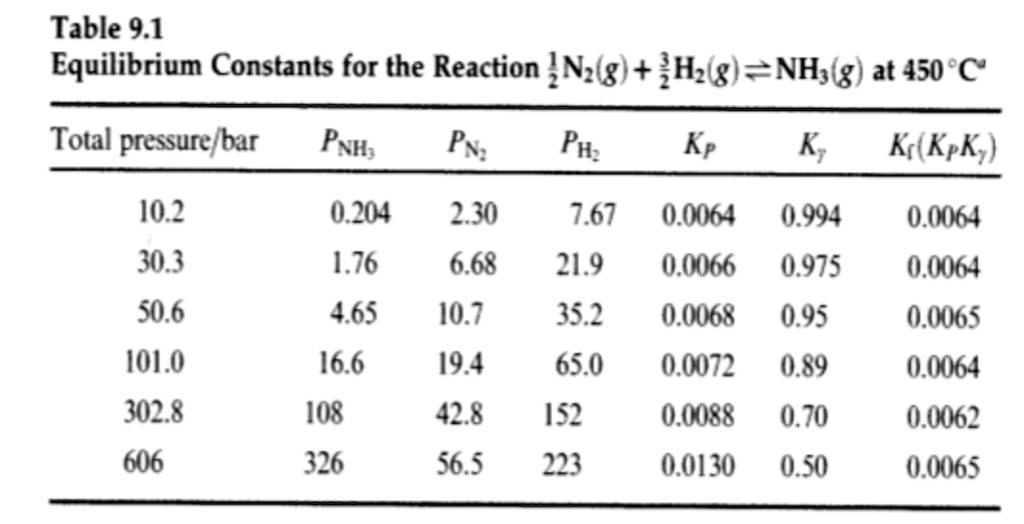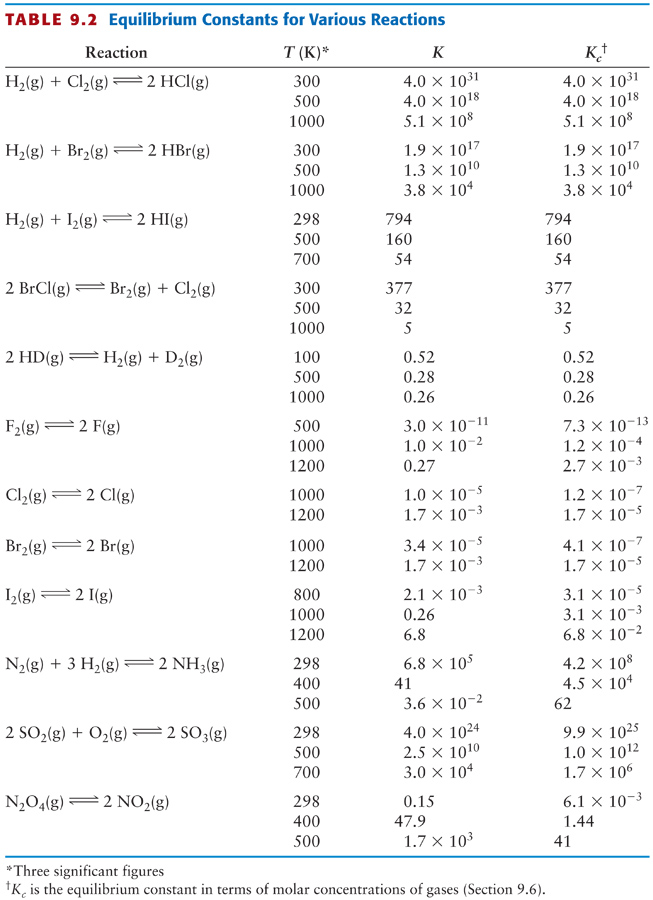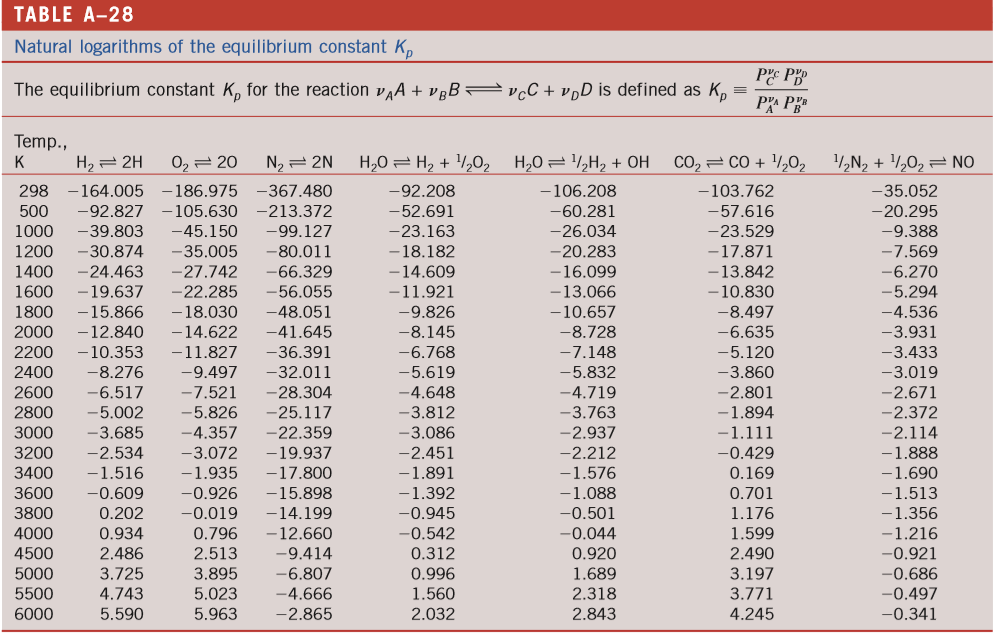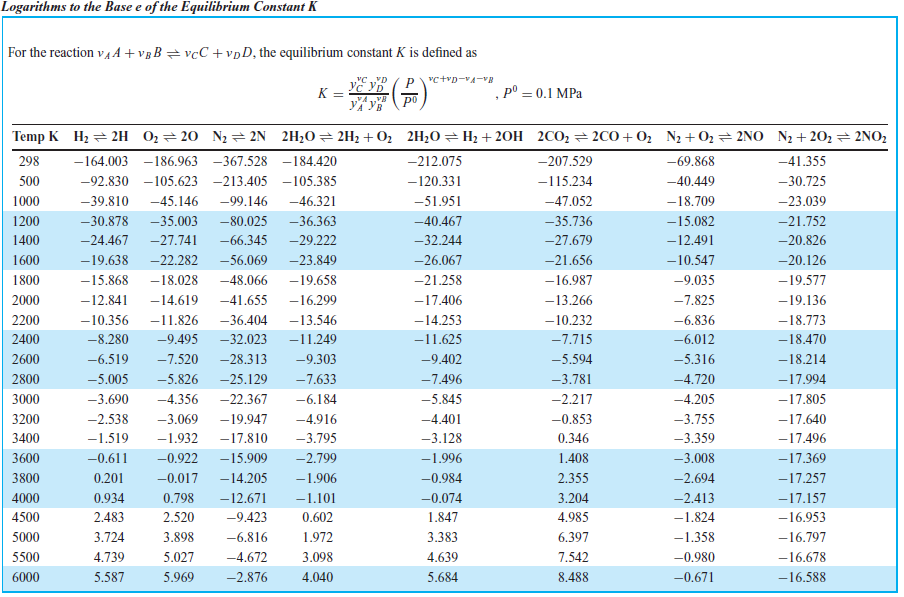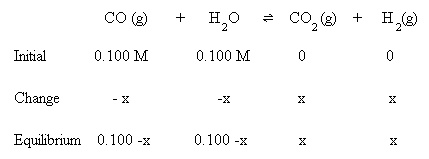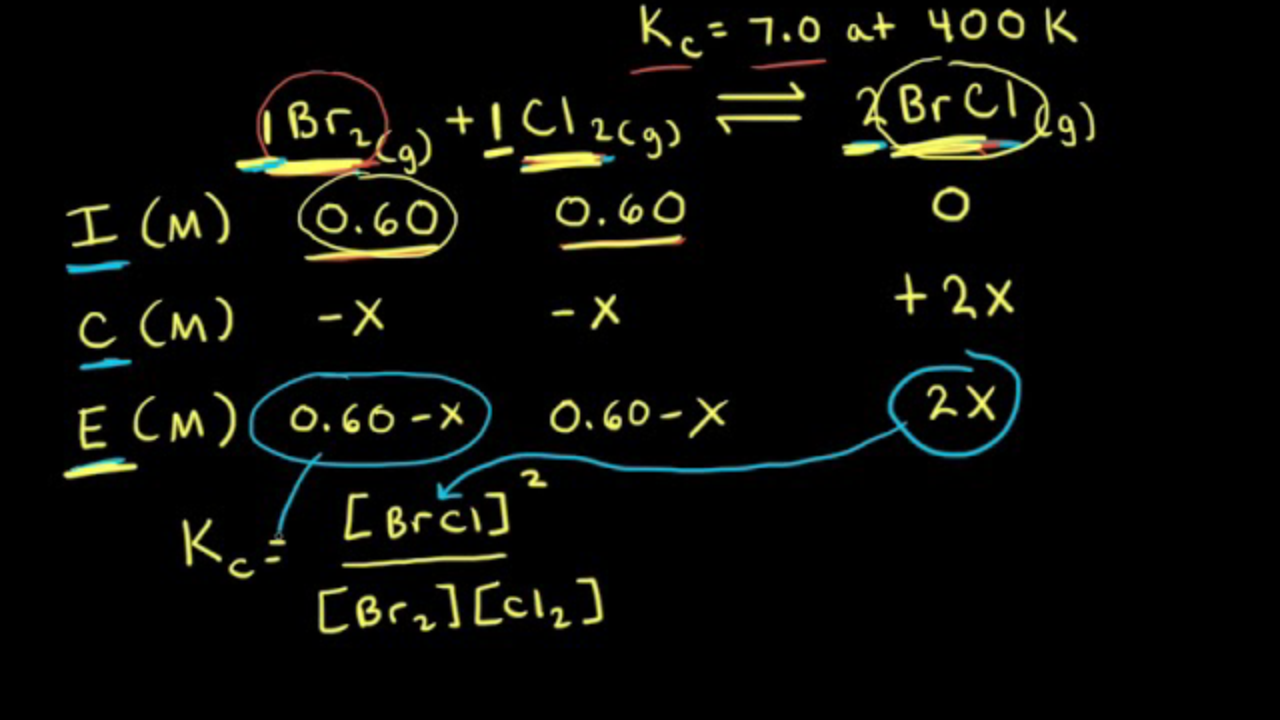
Calculating equilibrium concentrations from initial concentrations and the equilibrium constant (worked example) (video) | Khan Academy

Chemical Equilibrium - Ice Table - Equilibrium Constant Expression, Initial Concentration, Kp, Kc #5 - YouTube

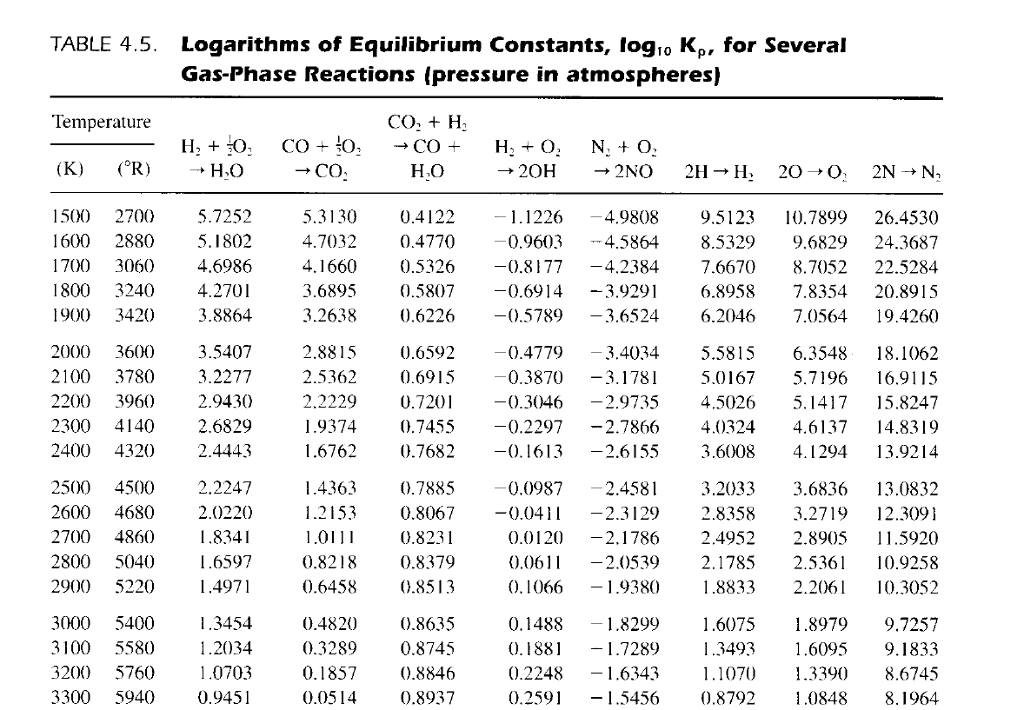
OneClass: Using this table of ionization constants, calculate the overall equilibrium constant, Kover...

Table 3 from Computational calculation of equilibrium constants: addition to carbonyl compounds. | Semantic Scholar

ICE Tables: Initial Concentration, Equilibrium Constant Expression - Chemical Equilibrium Problems. - YouTube